
Over the last few months, communities have come together to find solutions to the challenges driven by COVID-19. At Fitbit, we felt a strong sense of responsibility to support our community in the fight against COVID-19. So, when our co-founders challenged the team to find ways to leverage our existing expertise in advanced sensor technology, robotics, signal processing, supply chain logistics, hardware design, and more, and put them toward building solutions that could make a meaningful difference, a small team dove in on a secret project to help save lives.
After seeing an urgent need for ventilators around the globe, we saw an opportunity to respond based on our expertise. Enter Fitbit Flow, a high-quality, easy-to-use, and low-cost automatic resuscitator that is designed for emergency ventilation. It was inspired by the MIT E-Vent Design Toolbox and designed by Fitbit engineers and researchers, with valuable input from clinicians caring for COVID-19 patients from Oregon Health and Science Hospital and the MassGeneralBrigham Center for COVID Innovation on the needs of practitioners and, in particular, toward our still-evolving understanding of the needs of patients with COVID-19.
“COVID-19 has challenged all of us to push the boundaries of innovation and creativity, and use everything at our disposal to more rapidly develop products that support patients and the healthcare systems caring for them,” says James Park, co-founder and CEO of Fitbit. “We saw an opportunity to rally our expertise in advanced sensor development, manufacturing, and our global supply chain to address the critical and ongoing need for emergency ventilators and help make a difference in the fight against this global virus.”
We talked to two of the researchers on this project, Director of Research, Electrical Engineering Pete Richards and Senior Staff Research Scientist Dr. Tony Faranesh, to find out how the device works, why it’s important, and why the research team at Fitbit was well-positioned to create a device like this.
Development and testing
One of the jobs of the research team is to build things from beginning to end, including trying out new sensors and their functions, collecting the data, and understanding it. So when they took on the new challenge of developing an emergency ventilator, “it worked out pretty well to take some of the other things we’re familiar with and iterate quickly,” says Pete.
During development and testing, Fitbit consulted with doctors and clinicians from MassGeneralBrigham Center for COVID Innovation, as well as Drs. David Sheridan, Laura Chess, and Matthew Hansen, all in emergency medicine caring for COVID-19 patients at Oregon Health & Science University. San Francisco-based ICU physician Dr. Chris Tyler also provided a lot of helpful feedback around what capabilities would be essential and how a device like this fits into the bigger picture of current medical care for COVID-19 patients at hospitals.
With this crucial clinical feedback, the team at Fitbit was able to create a prototype based on a conventional manual resuscitator bag that paramedics use to manually resuscitate someone. Then, Fitbit Creative Director of Industrial Design Brian Paschke led the design team execution, Technical Director Jeffrey Borra the engineering effort, and Director of Manufacturing Engineering Sandeep Kadam the manufacturing effort.
The intention is to be able to use Fitbit Flow to support someone who needs mechanical ventilation when a commercial ventilator isn’t available. “We know from some conversations that physicians are already trying to work out the ethics in deciding who gets the ventilator and who doesn’t, due to shortage of supply,” says Tony. “The goal here is to support life in the event that one’s not available until one might become available.”
The design
According to the American Thoracic Society, a ventilator (also known as a mechanical ventilator, respirator, or breathing machine), is a life support treatment that helps people breathe when they can’t breathe on their own. The machine gets oxygen into the lungs and the body and helps to get rid of carbon dioxide through the lungs.
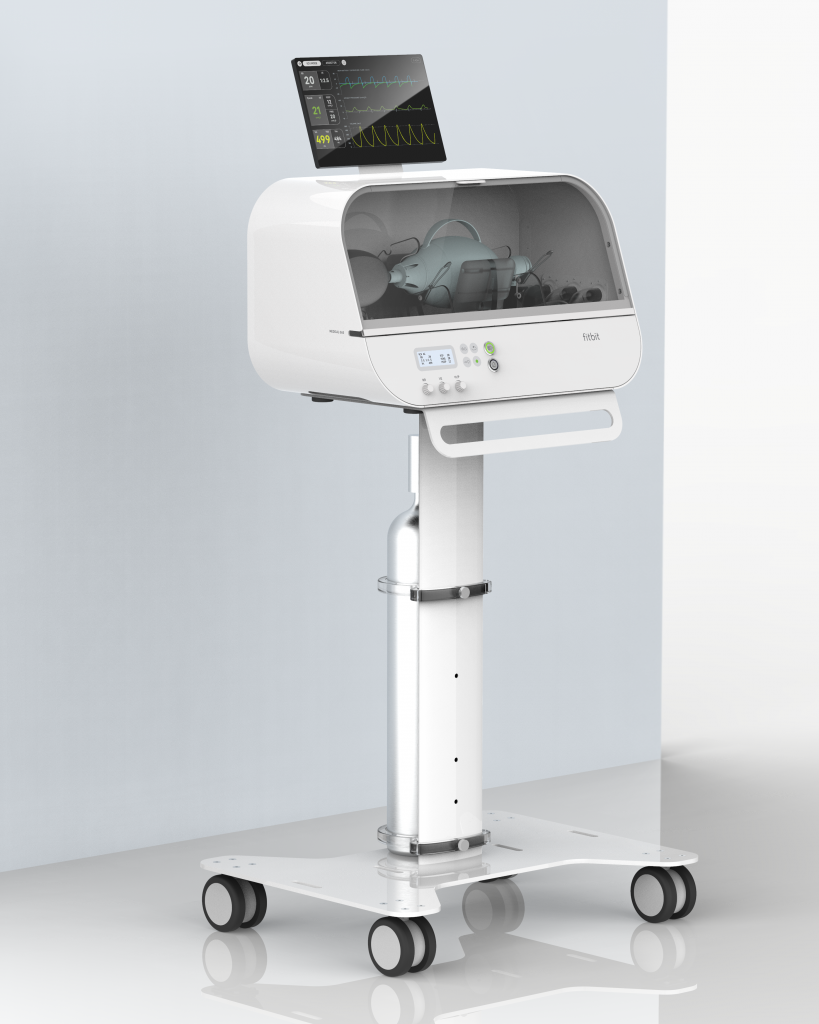
Fitbit Flow builds on standard resuscitator bags, like those used by paramedics, with sophisticated instruments, sensors, and alarms that work together to support patient monitoring and automated compressions with controlled delivery of volume or pressure.
Its form is intended to inspire confidence. Built from laser cut aluminum, it is designed to be intuitive and simple to use, potentially helping to reduce the strain on highly specialized staff who are typically needed to operate a commercial ventilator.
Plus, its hardware includes a clear viewing window for doctors to observe the automated pumping from a distance, and it can be used on a tabletop or with a stand for mobility. The stand is modular, with the ability to adapt to fit various size O2 tanks. The team continues to investigate opportunities to further improve the usability and capabilities of Fitbit Flow, including a plug-and-play, high-resolution add-on display, for viewing the system’s status and pressure/flow waveforms.
Creating a device that will help save lives—while working remotely
The biggest challenge in building the device was, of course, that most of the work had to be done remotely, as the team was, for the most part, under shelter-in-place lockdown just like the rest of us.
“It was an incredible team effort,” says Tony. “Commercial ventilators are incredibly sophisticated, nuanced machines. Based on limited information, the team had to build what they thought the most viable product was going to be that would be useful and helpful. People had to work around the clock, in isolation and just as individuals, to make decisions quickly, and still work together in these special circumstances. I’m proud of how quickly that came together.”
Fitbit has received Emergency Use Authorization from the FDA for use during the COVID-19 pandemic in situations where all of the available, conventional ventilators are not available. It aims to leverage the company’s vast infrastructure and manufacturing capabilities that currently produce millions of Fitbit devices per year to produce large volumes of these emergency devices quickly.
This information is for educational purposes only and is not intended as a substitute for medical diagnosis or treatment. You should not use this information to diagnose or treat a health problem or condition. Always check with your doctor before changing your diet, altering your sleep habits, taking supplements, or starting a new fitness routine.




If you have questions about a Fitbit tracker, product availability, or the status of your order, contact our Support Team or search the Fitbit Community for answers.
Please note: Comments are moderated and may not appear immediately after submission.